Our doctoral students and postdoctoral scholars are engaged in a variety of research projects aimed to elucidate how the olfactory system can subserve as a metabolic sensor.
Here is the 2022 recruitment poster for our lab. PDF
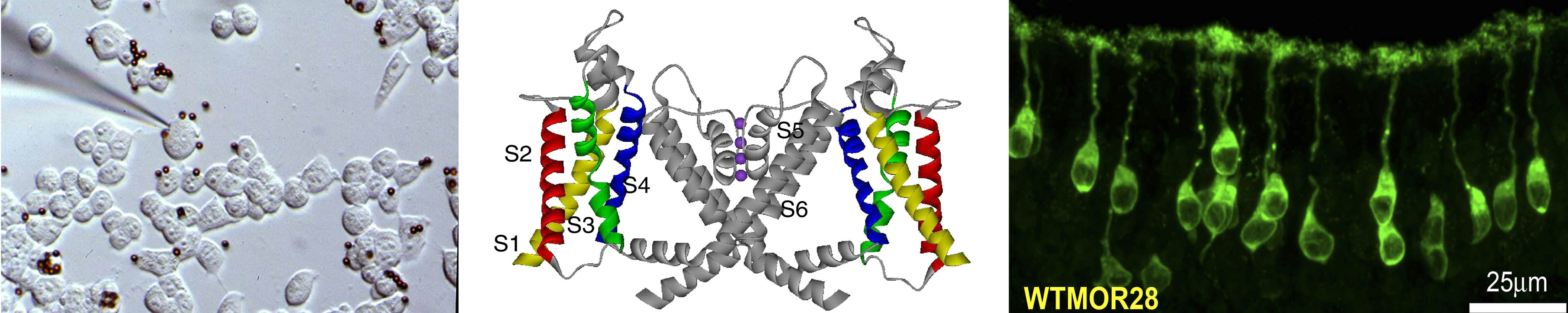
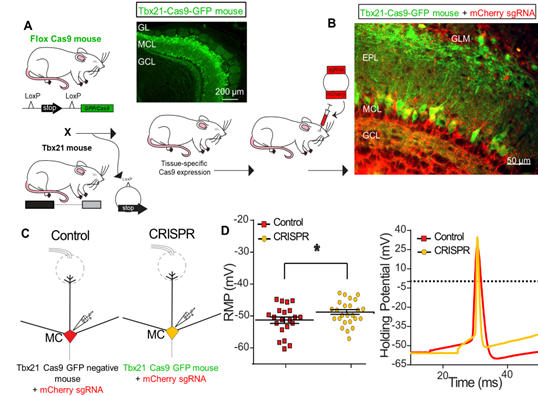
The Role of the Olfactory Bulb in Metabolism as Revealed by CRISPR (Louis Kolling)
We are exploring how energy homeostasis can be maintained by the olfactory system as a means to prevent diet-induced obesity and unwanted weight gain. I am using CRISPR gene editing and chemogenetics (called DREADDs) to up- and down-regulate output neurons of the olfactory bulb to modulate energy expenditure, adiposity, and weight gain when mice are challenged with a fatty diet. I explicitly used CRISPR to generate a conditional knockout of the potassium channel, Kv1.3, because these proteins regulate neuronal excitability and the resting membrane potential. I developed a technique to retro-orbitally deliver my designed CRISPR sgRNAs in AAV9 viral particles into genetically-engineered mice that express Cas9 in only mitral/tufted cells of the olfactory bulb. I used methodologies such as slice electrophysiology, odor habituation/dishabituation behavior assays, protein biochemistry, serum analysis, histology, and systems physiology (metabolic chambers) to characterize my CRISPR mice that demonstrated improved olfactory & health metrics compared to control littermates. I discovered that my engineered DREADDs mice oppositely demonstrated an obesogenic phenotype. Through my research, I have uniquely discovered that output neurons of the olfactory bulb communicate olfactory coding information and simultaneously participate in metabolic regulation.
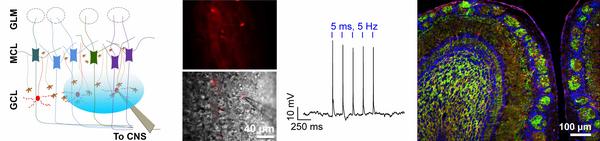
Gut to Brain Signaling or Local Neuromodulators of the Olfactory System? (Dr. Nicolas Thiebaud)
Although, we have known for a long time that appetite and olfactory perception are tightly interconnected, the exact mechanisms underlining the modulation of smell with the nutritional state remain poorly understood nowadays. The goal of our research is to identify how nutrients and hormones known to be involved in energy homeostasis can regulate the olfactory output at the olfactory bulb level. Using a combination of anatomical and electrophysiological tools, we study how mitral cell activity is regulated by key molecules such as insulin, glucose or the incretin hormone glucagon-like peptide 1 (GLP-1). I recently focused my research on GLP-1, a key regulator of energy metabolism and food intake, which has become an important therapeutic approach for treating type 2 diabetes. I developed an optogenetic approach using transgenic animal models to specifically activate a newly discovered population of GLP-1-producing interneurons in the olfactory bulb and revealed their modulatory action on mitral cells. Mitral cells are the main relay of the olfactory information between the olfactory sensory neurons in the nose and the higher brain areas responsible for processing the olfactory information.
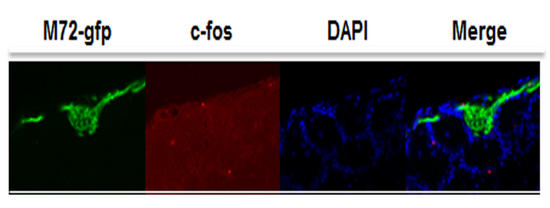
Mapping Odor-activated Neurons in Diet-induced Obese Mice (Dr. Erminia Fardone)
I am interested to understand changes in mapping odor-activated neurons in adult and middle-aged M72iresTaugfp transgenic mice treated with graded levels of diet-induced obesity (DIO). This work is based on the odor-induced immediate early gene expression, in particular c-fos, in the olfactory bulb of DIO transgenic mice. The main goal is to determine the extent and reversibility of olfactory sensory neuronal loss, reduced axonal targeting, and the plasticity of olfactory deficits. The work may lead to new knowledges on olfactory sensory neurons (OSNs) in response to high fat diet during aging.
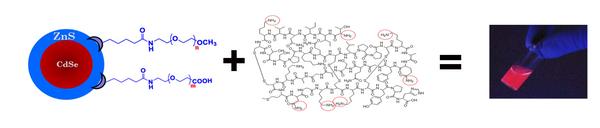
Nanoparticles as a Tool for Drug Delivery (Austin Schwartz)
Drug delivery is often hindered by the inability to selectively target a drug to a particular region of the brain or body as well as track the location of that drug following delivery. We have demonstrated that internal chemical state shapes olfactory ability and that electrical conductance of the olfactory bulb may drive whole body metabolism. The goal my dissertation is to bind drug molecules to nanoparticles and then to intranasally deliver this bioconjugate to the olfactory bulb (OB). These nanoparticles are either fluorescent (quantum dots) or magnetic (iron-oxide magnetic nanoparticles) and can be tracked following delivery ex-vivo through immunohistochemistry or in-vivo through magnetic resonance imaging (at the FSU National High Field Magnetic Laboratory). Following confirmation of targeting in the OB, I will later study the ability of these nanoparticle bioconjugates to alter physiological function in mice.
Serotonin is a Neuromodulator of the Main and Accessory Olfactory System (Zhenbo Huang, M.Sc.)
Serotonin (5-HT) is an important signaling molecule for various behavioral and physiological functions including mood, reward, aggression, appetite, and memory. 5-HT and its various receptors are widely distributed in the central nervous system. Here, I focus upon the serotonergic modulation in the olfactory system. More specifically, I compare neuromodulation of mitral cells by 5-HT in the main olfactory bulb (MOB) vs. that of the accessory olfactory bulb (AOB). I find that serotonergic modulation may be largely excitatory for the MOB mitral cells but predominantly inhibitory for the AOB mitral cells. The differentiated modulation of MOB and AOB by 5-HT may contribute to different behavioral regulations.

Can Exercise Obrogate the Detrimental Affects of Diet-Induced Obesity? (Brandon Chelette)
A hypokinetic lifestyle or consumption of an unbalanced, high-fat diet (or both) causes excess fat to be deposited resulting in weight gain. However, the detrimental effects of energy imbalance are not limited to superficial consequences such as needing to buy larger pants. Obesity is a high-risk disease that is correlated with cardiovascular disease, diabetes, cognitive deficits, and other maladies. Our lab’s recent findings have shown that the damage caused by metabolic imbalance even extends to the olfactory system. Mice maintained on a high-fat diet gain weight, of course, but they also lose important neurons in the olfactory bulb and have decreased odor-learning capabilities. The question is can we combat the negative effects of a high-fat diet in mice by simulating an active lifestyle? The physiological benefits of exercise are well documented, and again extend beyond cosmetic effects like weight loss. Consistent exercise reduces body fat, but also increases cognitive function, reduces inflammation, and stimulates neurogenesis in the hippocampus. My research focuses on using voluntary exercise and diet manipulation in an attempt to 1) ameliorate the negative effects of a high-fat diet and 2) investigate neuromodulation in the olfactory system of normal and transgenic mice.

Fatty Diet-induced Neuroinflammation (ashley loeven)
Our research has shown that mice fed a moderately high-fat diet exhibit a loss of olfactory sensory neurons. I have found that mice that are isocalorically matched on a diet that prevents obesity but contains 32% fat, still exhibit neuronal loss in the olfactory epithelium. This narrowed down my research focus from diet-induced obesity to dietary fat. The molecular process behind this loss is unknown, and the irreversible nature of the neuronal loss is interesting to me. It is known that dietary fat leads to whole-body inflammation and gut microbiome changes. I suspect that the gut microbiome shift induces systemic inflammation, which leads to an inflammatory state of the olfactory system and that this is responsible for the imbalance in neuroregeneration and cell death. I am exploring the connectivity of dietary fat, the gut microbiome, circulating inflammatory factors, neuroinflammation, and olfactory sensory neuronal loss.
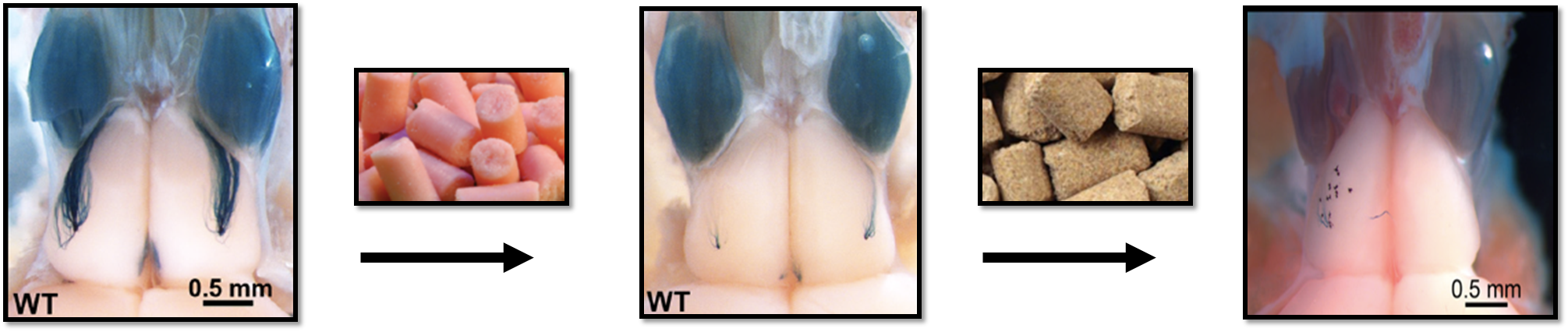
Systemic Metabolic Changes as a Result of Ablating Olfactory Sensory Neurons in the Main Olfactory Epithelium and the Bulb (Destinee Gatlin)
Chemosensory cues help animals detect changes in their environments to assist with reproduction and survival. Previously, our lab discovered that diet-induced obesity causes an inadequate ability to detect chemosensory signals due to a malfunctioning in olfactory sensors and overall loss of olfactory sensory neurons. However, the link between the subsequent loss of olfactory sensory neurons and metabolism has yet to be understood. My project evaluates metabolic effects of olfactory sensory neuron ablation in the main olfactory epithelium using methimazole, unilateral naris occlusion and genome editing. Comprehensive Lab Monitoring System (CLAMSTM) chambers and behavioral assays such as the buried cookie assay were used to facilitate data collection. Our lab has also previously discovered that excess nutrition leads to a decrease in total energy expenditure. Thus, we hypothesize that the manipulation of olfactory sensory neurons might result in similar decreases in energy expenditure.
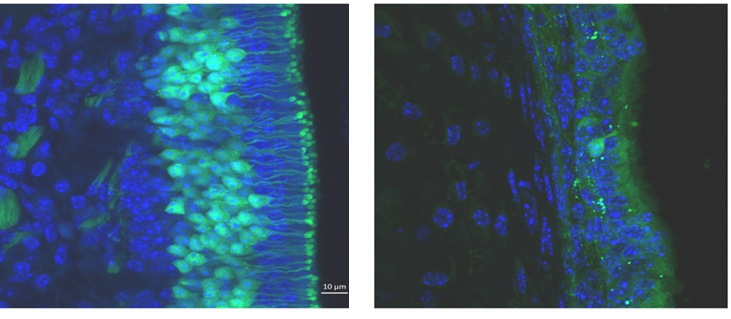
Neurogenesis, Restricted Eating, and Diet-induced Obesity (Ryan Ochoa)
Adult neurogenesis occurs in the mammalian olfactory bulb throughout life and is essential for olfactory learning and processing. It is known that the survival of newly born neurons is dependent on synaptic integration from locally established cells in the olfactory bulb. Previous research shows that this synaptic integration is prompted by olfactory related activity within the bulb; few studies show the relationship between metabolic signals and neurogenesis in the olfactory bulb. Given the wide variety of satiety and hunger proteins in the olfactory system, and the detrimental effects diet-induced obesity has on it, I decided to focus my research on the link between hunger and satiety signals and neurogenesis in the olfactory bulb. Using restricted feeding and diet-induced obesity models I can control nutrient availability to determine the role of metabolic signaling in protein secretion, protein conformation changes and excitability in the bulb, then relate these to anatomical changes in the olfactory system.