Research Overview: Olfactory Signaling and Neuromodulation of Ion Channels
We are researching learning, memory, and neural plasticity at the level of the ion channel protein. Our main stay in the laboratory is biophysics, specifically a technique called patch-clamp electrophysiology, where we can measure single conformational changes in ion channel proteins that elicit electrical signals, essentially the language of the brain. One of the most ubiquitous ways of modulating electrical activity of ion channels is a biochemical process called phosphorylation, whereby negative phosphate groups are added to the channel at specific residues. Hence, we combine our skills in electrophysiology with those of protein biochemistry (phosphorylation assays; protein-protein interactions), molecular biology (creating mutant ion channels and signaling proteins), and molecular genetics (genetically targeted "knock-out" mice to study cell signaling by loss of function).
We were very much excited that the 2000 Nobel Prize in Physiology or Medicine was attributed to several scientists that discovered the importance of phosphorylation. Perhaps the importance of phosphorylation in regulating cellular activity can be underscored by the large portion (2-3%) of the eukaryotic genome set aside to code for kinases, enzymes that initiate phosphorylation. Humans have 2000 conventional kinase genes and most of those exist in the brain. Abnormality in these genes and correlate enzyme activity could contribute to the onset or severity of specific neuronal diseases such as Alzheimer's functional, inflammatory responses, deregulated cell proliferation, and to diseases such as cancer (especially mammary), atherosclerosis, psoriasis, and diabetes.
We have discovered that hormones and neurotrophins (glucagon-like peptide 1 (glp-1), insulin, and brain-derived neurotrophic factor (BDNF)) modulate electrical activity in the brain at the level of the ion channel via phosphorylation. These types of signaling molecules are also involved in maintaining energy homeostasis. Currently, we hypothesize that the olfactory system not only senses our external chemical environment (odors) but is a detector of internal chemistry, the chemistry of metabolism. As such, we are studying the neuropathology of diabetes and obesity - how sensory systems, such as the olfactory system are perturbed by diet-induced obesity at the level of the ion channel.
I would welcome students wishing to gain experience with the techniques of electrophysiology (patch-clamp recording, optogenetics, and single channel), protein biochemistry, molecular biology (site-directed mutagenesis, gene-targeted deletion, DREADS, and viral infection), immunocytochemistry (neural activity markers), cell biology (cell culture, ELISA), olfactometry (computerized Knosys), systems physiology (CLAMS metabolic chambers), and mouse behavioral phenotyping (two-choice paradigm, odor habituation/dishabituation, elevated-plus maze, T-maze), and to those wishing to answer questions about the physiology of olfaction, cell-signaling cascades, the regulation of neuronal excitability, gut to brain metabolic processes, and ion channel structure/function.
Obesity and the Brain
The electrical signals of the olfactory system are reduced by diet-induced obesity. Consumption of excess energy substrates affects all the organ systems of your body - and your brain is not immune.
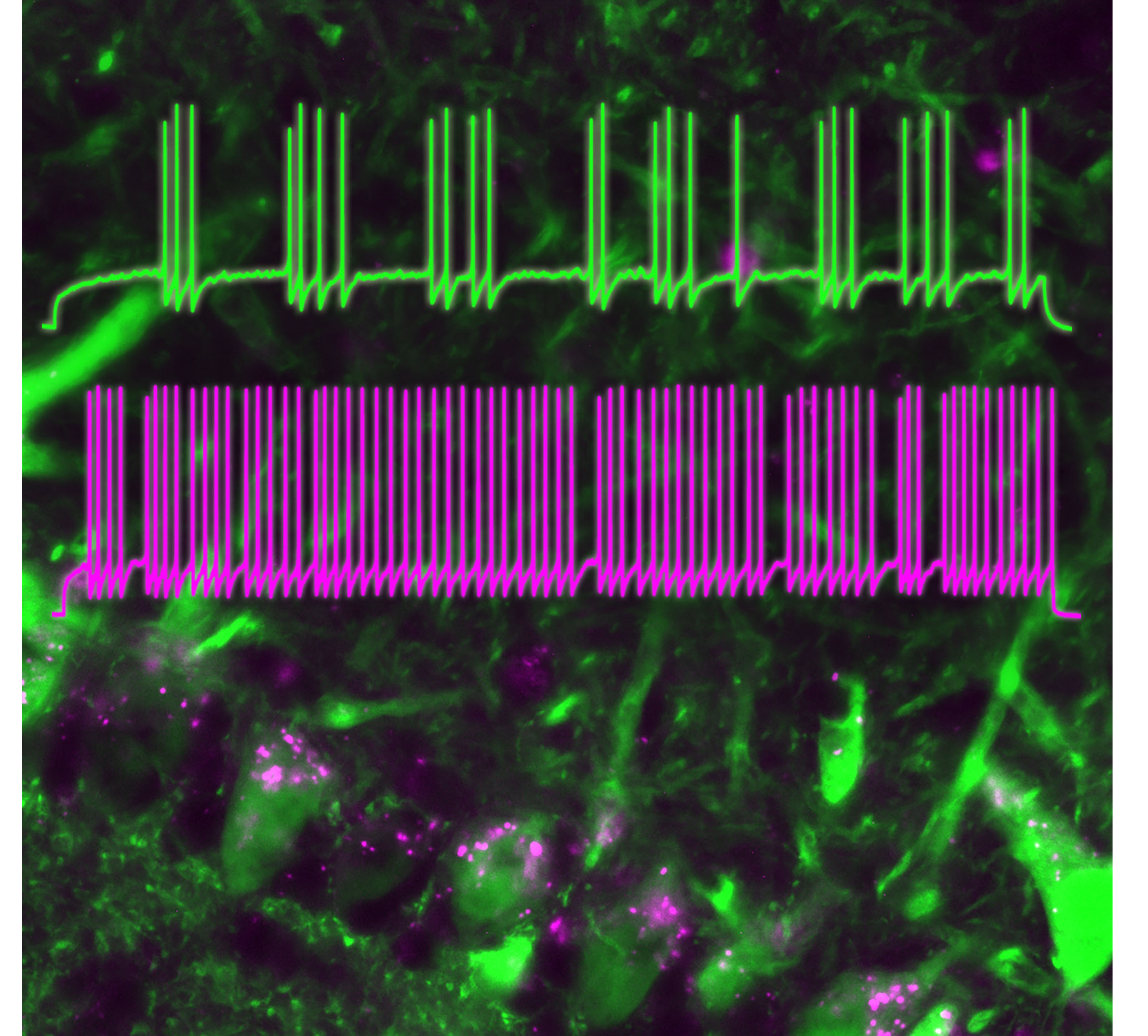
Techniques and Experimental Approaches
We know that "the thinking brain passes around electrical signals generated by a single class of proteins, the ion channel." (Christopher Miller, 1990) Our main stay in the laboratory is biophysics, specifically a technique called patch-clamp electrophysiology, where we can measure single conformational changes in ion channel proteins that elicit electrical signals, essentially the language of the brain.
Resources
We use a number of automated, technical, or custom-engineered instruments in the laboratory to collect information about olfactory ability, learned behaviors, whole-system metabolism, ingestive behavior, electrical signaling, or anatomical features.